Vol 14, No 1 (2019)
View or download the full issue
PDF (Russian)
CHEMISTRY AND TECHNOLOGY OF MEDICINAL COMPOUNDS AND BIOLOGICALLY ACTIVE SUBSTANCES
5-24 12834
Abstract
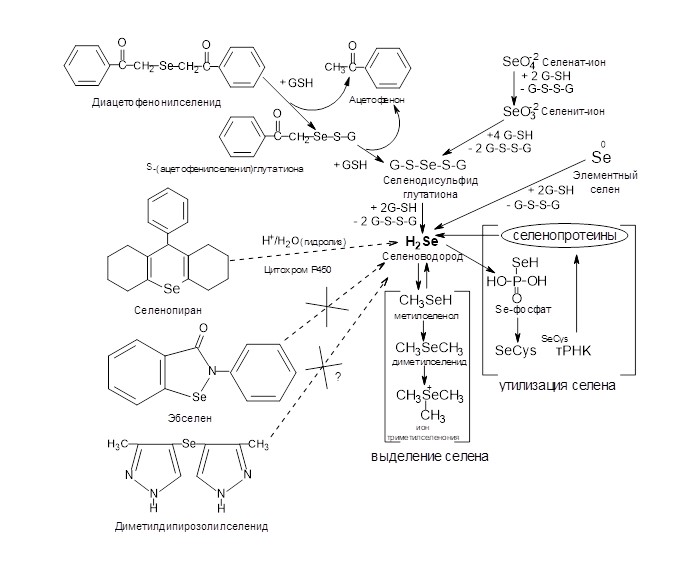
The work presents a review devoted to the metabolism and the mechanism of toxicity of seleniumcontaining supplements: elemental selenium, sodium selenite, diacetophenonyl selenide, selenopyrane, ebselen, dimethyl dipyrasolyl selenide and selenium-containing amino acids used for correction of selenium deficiency. Elemental selenium penetrating through cell walls, but not through transport channels demonstrates poorly predicted and difficultly regulated bioavailability. Sodium selenate is known to be the most toxic form of selenium in food. The metabolism of xenobiotic diacetophenonyl selenide resembles that of sodium selenide. The xenobiotic reacts with thiols, for instance, with the reduced form of glutathione leading to the formation of hydrogen selenide. Ebselen is not considered to be a well bioavailable form of selenium and thus possesses low toxicity. Xenobiotic selenopyrane eliminates selenium only in processes of xenobiotic liver exchange, and in our investigations - partially in acid-catalyzed hydrolysis. The metabolism of xenobiotic dimethyl dipyrasolyl selenide having low toxicity is poorly investigated. The toxicity of high doses of selenomethionine is determined by the possibility of incorporation in proteins and vitally important enzymes with dramatic changes of protein quaternary structure. The toxicity of high doses of methylselenocysteine seems to be caused by the lack of an exchange pool in the body and quick regeneration of hydrogen selenide from methylselenol which is formed as a result of enzymatic destruction of this amino acid. Also the issue of the most prospect selenium donor is discussed. The physiological compatibility, the low toxicity, the presence of an exchangeable pool in the organism, the antioxidantal properties and the simplicity of production indicate selenocystine as an optimal selenium donor.
25-31 1216
Abstract
The article investigates the aqueous solutions of food chitosan and glutaraldehyde to determine the feasibility of their use as components of an embolizing composition. It was shown on the basis of experimental flow curves, viscosity and velocity curves that test solutions have low viscosity and exhibit Newtonian flow behavior. The activation energy of viscous flow of the fluids was estimated in the temperature range of 25-37 °C within the Arrhenius - Frenkel - Eyring equation. It varies within a narrow range: 17-24 kJ/mol. When mixing the aqueous solutions of food chitosan and glutaraldehyde, chemical interaction of the solutes occurs. It is accompanied by an increase in viscosity and formation of a covalently crosslinked gel. Using a simple exponential equation the effective rate constant of the chemical process was calculated. It varies in a wide range: 1.9-82.7∙103 1/s. These values can be used when selecting an optimal region of food chitosan-glutaraldehyde ratios and concentrations of their aqueous solutions to generate embolizing agents. The conditions at which gel formation takes place over forty seconds were determined. Differential scanning calorimetry indicated a negligible thermal effect of food chitosan reaction with glutaraldehyde in the aqueous medium, which ensures no thermal burn during the formation of an embolus in the blood vessel in situ. As a result of the work elastic solid gels suitable for use as embolizing agents were obtained.
32-38 1125
Abstract
Photooxidized psoralen solutions possess a variety of biological effects, which implementation mechanism may presumably involve hydroperoxides. Here, the hydroperoxide content in photooxidized psoralen solutions was assessed using photometric FOX assay (from Ferrous Oxidation + Xylenol Orange). FOX reagent with 10× content of Xylenol Orange, modified for quantitative analysis of up to 50 μM of hydroperoxides in aqueous phase was used in experiments. During photooxidation of 0.1 mM psoralen in phosphate buffer solution, hydroperoxide production increases with dose of UVA irradiation (~2.5 μM eq. of H2O2 for dose of 252 kJ/m2 and ~11 μM eq. of H2O2 for dose of 1512 kJ/m2) and reaches ~16.5 μM eq. of H2O2 at the highest dose investigated (3024 kJ/m2). A comparison of kinetics of psoralen photolysis and hydroperoxide generation allows us to suggest that generation of hydroperoxide results from the secondary photochemical processes involving psoralen photoproducts, presumably from photoinduced autooxidation of aldehydic photoproducts of psoralen.
THEORETICAL BASIS OF CHEMICAL TECHNOLOGY
39-46 1399
Abstract
The formalized model of carbon sorbent synthesis control based on the methodology for functional modeling is constructed. The correlations between the directions of use and the properties of carbon sorbents are revealed. The characteristics that are essential regardless of the direction of use of the sorbent, in particular, sorption properties and strength are identified. The technologies based on the gas-phase method of obtaining carbon material are considered, the analysis of individual stages of the process of obtaining carbon sorbents is carried out. The analysis of the influence of the technological parameters of the synthesis on the properties of sorbents is carried out. On the basis of the established relationships, a functional model has been built that provides a hierarchically ordered, structured, visual description of the management of carbon sorbent synthesis. The simulation is performed “from top to bottom” from the most general description to the detail. The resulting model is a set of interrelated graphical diagrams. At the initial stage, the synthesis of carbon sorbent is considered as a single process, the input parameters of which are hydrocarbon gas, the activating agent and the material form factor, the output - carbon sorbent, and the control parameters are the requirements for strength and sorption properties. Then the synthesis process is decomposed. The control processes (analysis of raw material properties and matrix selection), technological processes (raw material preparation) and mixed processes are distinguished as a result of decomposition. The model includes a consistent description of the technological parameters selection (temperature, gas flow and time) for both stages of the synthesis process. The model is the base for information support providing for the production of carbon sorbents with the required properties.
47-58 1757
Abstract
New experimental data on the chemical equilibrium in the propionic acid - ethanol - ethyl propionate - water system at 293.15 K and atmospheric pressure are presented. Chemically equilibrium compositions corresponding to the liquid-liquid equilibrium were obtained by gas chromatographic analysis. Using the method of nuclear magnetic resonance, homogeneous chemically equilibrium compositions were determined and the concentration equilibrium constant is calculated. The surface of chemical equilibrium and the region of splitting chemically equilibrium compositions are represented in the square of the transformed concentration variables. Comparison of the data obtained in the work with the literature was carried out at 303.15 and 313.15 K. It was found that the region of such compositions decreases with increasing temperature, while the surface of chemical equilibrium does not change the shape and position in the concentration space in the temperature range 293.15-313.15 K and atmospheric pressure. Liquid-liquid equilibrium compositions have also been obtained by gas chromatographic analysis for ethanol and ethyl propionate in the pseudo-ternary system using deep eutectic solvents (DES) based on choline chloride and glycerol / urea in whole range of concentration. The analysis of the extraction properties of DES showed the highest efficiency of DES based on choline chloride and urea. Experimental data on phase equilibrium are processed using Othmer-Tobias and Hand models. The calculated correlation coefficient (more than 0.99) indicates a high internal consistency of the experimental data obtained in this work.
CHEMISTRY AND TECHNOLOGY OF INORGANIC MATERIALS
59-65 1055
Abstract
This paper describes the method for obtaining a new derivative of the closo-decaborate anion with diethylenetriaminepentaacetic acid (DTPA) as a pendant group attached to the boron cluster through an alkoxyl spacer chain. This derivative is formed by the interaction of 1,4-dioxane derivative of the anion [B10H10]2- with DTPA potassium salt in an aqueous medium. As a result of the reaction, an exo-polyhedral cyclic substituent is opened, and then the addition of a polyfunctional group through an oxygen atom occurs. The synthesized compound is in fact an effective polydentate ligand capable of coordinating to the complexing agent both due to the donor atoms of the attached DTPA fragment and through the formation of three-center two-electron bonds. The obtained compound interacts with gadolinium(III) carbonate forming a complex of the composition [Gd2B10H9O2C4H8(dtpa)]·3H2O. The synthesized substances were studied by IR spectroscopy, polynuclear (11B, 13C and 1H) NMR spectroscopy, ESI mass spectrometry, elemental and thermographic analysis. closo-Decaborate with the pendant DTPA group is of interest in 10B neutron capture therapy of malignant tumors due to the high content of boron atoms and a convenient way of their transport to the affected cells. The obtained boron-containing derivatives of gadolinium(III) can act as drugs of combined action, because they can perform, in addition to the above described therapeutic function, the diagnostic function due to the presence of gadolinium atoms int hem.
CHEMISTRY AND TECHNOLOGY OF ORGANIC SUBSTANCES
66-74 1667
Abstract
Polyhydric alcohols - neopolyols and their derivatives due to the nature of the structure have a row of unique properties - high thermal stability, moisture resistance, chemical resistance and are widely used in the production of synthetic oils, resins, varnishes, surfactants and plasticizers. Esters of neopolyols with several ester groups have excellent characteristics at low temperatures, as well as high environmental friendliness, which meets modern requirements. The variation in the production of esters of neopolyols, alcohol and acid components in various combinations allows to obtain a wide range of products with different characteristics and consumer properties. Currently, neopolyol esters are not produced in Russia. However, the technical capabilities of the development of R-Oxo technology based on natural gas are not in doubt, which will allow in the near future to organize its own production of neopolyol esters for the production of high-tech materials. In the present work, the possibility of obtaining plasticizers based on trimethylolpropane esters and C2-C5 acids of various structures was investigated. Samples of 7 trimethylolpropane triesters were synthesized and their certain physicochemical properties were determined. It is shown that the most promising is the synthesis of a plasticizer using acetic acid. Conducted kinetic studies on the esterification of trimethylolpropane with acetic acid. A differential method was used to estimate the reaction rate from the initial consumption rates of acetic acid. The first orders of reaction are determined by alcohol and catalyst under the conditions studied; the value of the observed activation energy of the obtained reaction of the pseudo-second order, in the temperature range of 80-115 °C, was 57.9±8.1 kJ/mol. The results obtained allow us to recommend the conditions for the implementation of the process in the industry.
75-81 977
Abstract
The research results of the influence of preliminary mechanochemical activation of heavy oil feedstock (oil-fuel, tar) on the output of their coking products are presented. Raw materials were activated by creating a cavitation effect when a hydrocarbon stream passes through a diffuser under a pressure. The pressure gradient on the diffuser ranged from 10 to 50 MPa, and the number of cycles of passage through the diffuser - from 1 to 5. It is shown that hydrodynamic cavitation processing results in a change in the physicochemical characteristics of heavy oil feedstock. The presented values of the raw material density and its fractional composition before and after processing suggest that the cavitation effect results in cracking reactions in the raw material. Changes in the characteristics of the raw material affect the change in the output of its coking products. It is established that as the pressure gradient and the number of impact acts increase, the yield of liquid coking products increases, and the yield of coke decreases. No relationship between the feedstock characteristics and the increase in the yield of distillate fractions as a result of prior cavitation effects has been established. At the same time, the preliminary mechanochemical activation of the raw materials leads to a decrease in the density and refractive index of the liquid coking products, a decrease in the temperature of their initial boiling point, and an increase in the content of hydrocarbons in the gasoline and kerosene fractions in them. The increase in the density and refractive index of the light fractions of the liquid coking products suggests that the aromatization process proceeds as a result of mechanochemical activation.
SYNTHESIS AND PROCESSING OF POLYMERS AND POLYMERIC COMPOSITES
82-89 1175
Abstract
The possibility of synthesizing silatrane-containing polymers was investigated using three different synthetic methods: the formation of silatrane fragments from polymers with trialkoxysilyl groups, the copolymerization of silatrane-containing monomers, and the reaction of silatranes with functional copolymers. The obtained polymethacrylate copolymers were characterized using gel permeation chromatography, IR and NMR spectroscopy. It was shown that depending on the synthesis scheme used, polymers were obtained in the form of three-dimensional structures or soluble products. It was established that the molecular weight of the synthesized polymers depended significantly on both the content of silatrane fragments and the synthesis technique used. It was shown that the modification of linear carboxyl-containing copolymers by silatranes allows the synthesis of high-molecular polymers with a high content of silatrane fragments. For the synthesized polymers, thermal properties were investigated, and the hydrophobicity of the surface of polymer films was also evaluated. It was found that all the studied polymers did not have clear melting and crystallization temperatures. The polymers were stable in an inert atmosphere up to 270-280 °C, whereas in air they decomposed at lower temperatures with the restructuring of the macromolecular skeleton and the formation of highly heat-resistant silicone structures. An increase in the content of silatrane moieties in the copolymers led to an increase in the hydrophilicity of polymers.
P. V. Surikov,
N. L. Shembel,
A. A. Yurkin,
A. V. Petrogradsky,
V. D. Sevruk,
I. D. Simonov-Emelyanov
90-96 1025
Abstract
By the method of capillary viscosimetry, the melt flow curves of the molded phenolic resin and its compositions with lubricant, plasticizer and their mixture were obtained. It was shown that the size (diameter) of the capillary channel influences the dependence of the effective shear rate on the shear stress (flow curves) of the studied compositions. Such rheological behavior of the compositions during flow is associated with the effect of sliding along the surface of the capillary wall. According to the Mooney method, the dependences of the effective shear rate at given values of shear stress on the reciprocal of the capillary radius are plotted. The function of the slip velocity on the shear stress on the capillary wall is characterized in terms of the slip coefficient, which relates the shear stress on the capillary wall to the velocity of the composition along it. For the studied compositions, the total flow through the capillary was divided into volume fractions, one of which is associated with a shear flow; the other is determined by the slip effect. It has been shown that the introduction of both a lubricant and a plasticizer into the composition leads to an increase in the fluidity of the compositions. At the same time, adding of lubricant increases the volume fraction of the slip flow. The greatest effect of increasing the fluidity of the composition gives the use of complex modifying additives containing both lubricant and plasticizer.
ISSN 2410-6593 (Print)
ISSN 2686-7575 (Online)
ISSN 2686-7575 (Online)