THEORETICAL BASIS OF CHEMICAL TECHNOLOGY
- Different types of distillation (i.e., direct, indirect, sharp) were analyzed for the separation of quaternary mixtures containing components exhibiting similar volatility.
- The areas of energy advantage for flowsheets based on the use of the direct, indirect, and sharp distillation were allocated in composition tetrahedron.
- Recommendations on the choice of one distillation type for the separation of quaternary mixtures were formulated.
Objectives. The conditions for the effective application of the sharp distillation technique (without a component distributed between the distillate and bottom flows) for the separation of quaternary zeotropic mixtures containing components with similar volatilities were determined. The area of energy advantage for the flowsheet based on the preliminary fractionation of the mixture, compared with the flowsheet, the first distillation column of which works based on the indirect separation mode, was identified for an ethyl acetate–benzene–toluene–butyl acetate system. Energy savings of up to 20% were achieved. The direct and indirect distillation modes can become competitive when the point of the original composition is located near single K-surfaces or in a region with a different ratio of distribution coefficients. Sharp distillation is not suitable for the separation of a mixture containing a pair of components exhibiting relative unity volatility with medium boiling points.
Methods. The mathematical modeling in the Aspen Plus V.10.0 software package was chosen as the research method. The simulation was based on the Wilson local composition equation. The relative errors in the description of the phase equilibrium did not exceed 3%.
Results. The structure of the vapor–liquid equilibrium diagram and diagram of surfaces of the unit component distribution coefficients were studied for the ethyl acetate–benzene–toluenebutyl acetate and acetone–toluene–butyl acetate–o-xylene systems. Flowsheets based on the sharp, indirect (both systems), or direct (second system) distillation modes were proposed. The distillation process was simulated, and the parameters of the column work were determined (the quality of the substances meets the State Standard requirements of the Russian Federation for minimal energy consumption).
Conclusions. Recommendations regarding the use of sharp distillation for the separation of quaternary mixtures containing components with similar volatilities were devised.
The recovery of valuable compounds from aqueous mixtures of volatile organic solvents can be achieved utilizing extractive distillation techniques. For this purpose, the use of selective and non-toxic agents is preferred. For example, isolation of tetrahydrofuran from its ternary mixtures with different compositions of methanol and water is possible using glycerol.
Fig.: A – glycerol, 1 – methanol, 2 –tetrahydrofuran, 3 – water.Objectives. Synthesis and comparative analysis of the extractive distillation flowsheets for aqueous mixtures of solvents utilized in pharmaceutical industries using the example of a methanol−tetrahydrofuran−water system with various compositions. The ternary system contains two minimally boiling azeotropes that exist in a vapor–liquid phase equilibrium. To evaluate the selective effect of glycerol, the phase equilibria of the methanol–tetrahydrofuran–water and methanol–tetrahydrofuran–water–glycerol systems at 101.32 kPa were studied.
Methods. The calculations were carried out in the Aspen Plus V.9.0 software package. The vapor–liquid equilibria were simulated using the non-random two-liquid (NRTL) equation with the binary interaction parameters of the software package database. To account for the non-ideal behavior of the vapor phase, the Redlich–Kwong equation of state was used. The calculations of the extractive distillation schemes were carried out at 101.32 kPa.
Results. The conceptual flowsheets of extractive distillation are proposed. The flowsheets consist of three (schemes I–III) or four (scheme IV) distillation columns operating at atmospheric pressure. In schemes I and II, the extractive distillation of the mixtures is carried out with tetrahydrofuran isolation occurring in the distillate stream. Further separation in the schemes differs in the order of glycerol isolation: in the third column for scheme I (traditional extractive distillation complex) or in the second column for scheme II (two-column extractive distillation complex + methanol/water separation column). Sсheme III caters to the complete dehydration of the basic ternary mixtures, followed by the extractive distillation of the azeotropic methanol–tetrahydrofuran system, also with glycerol. Sсheme IV includes a preconcentration column (for the partial removal of water) and a traditional extractive distillation complex.
Conclusions. According to the criterion of least energy consumption for separation (the total load of the reboilers of distillation columns), sсheme I (a traditional complex of extractive distillation) is recommended. Additionally, the energy expended for the separation of the basic equimolar mixture using glycerol as the extractive agent was compared with that expended using another selective agent: 1,2-ethanediol. Glycerol is an effective extractive agent because it reduces energy consumption, in comparison with 1,2-ethanediol, by more than 5%.
CHEMISTRY AND TECHNOLOGY OF MEDICINAL COMPOUNDS AND BIOLOGICALLY ACTIVE SUBSTANCES
- An alternative method for the purification of a guanidine derivative with promising antiseptic properties has been proposed.
- The specified purification method results in a reduction in the content of residual monomers.
- The described purification method enables synthesis of branched hydrosuccinate oligohexamethylene guanidine exhibiting high purity, which meets the standards of the 14th edition State Pharmacopeia of the Russian Federation.
Objectives. Given that microorganisms can become resistant to certain groups of drugs and considering also their ability to form biofilms, the development of new drugs that are active against adapted microflora is required. This study focused on the development of a new method for the synthesis of a promising compound, the branched hydrosuccinate oligohexamethylene guanidine (OHMGsucc), with high purity that meets the standards of the 14th edition State Pharmacopeia of the Russian Federation (SPRF). Previously proposed methods have managed to isolate this product, which, however, complies with the requirements of the outdated SPRF. Therefore, the main aim of this study was to update the regulatory framework for the indicated OHMG salt for its further use in the pharmaceutical industry according to modern standards.
Methods. To control the residual impurities of hexamethylenediamine (HMDA) and guanidine hydrochloride (GHC), high-performance liquid chromatography (HPLC) was applied using a Thermo Scientific Dionex UltiMate 3000 chromatograph, and the chromatographic signals of the test solution with those of a standard sample solution obtained by a previously published conventional method were compared.
Results. The HPLC experimental data indicated a significant difference in the quantitative content of HMDA and GHC observed for the new and older preparation method of the branched OHMGsucc, suggesting that the method disclosed in this article can be used to obtain highly pure OHMGsucc.
Conclusions. The specified compound was standardized with the parameter “related impurities” according to the current (14th) edition of the SPRF. The effectiveness and reproducibility of the proposed method was experimentally confirmed. In addition, a process diagram for the preparation of the indicated OHMG salt was prepared.
The flavonoid profile of hypoglycemic collection (HGC) consisting of the leaves of the common bean (Phaseolus vulgaris L.), shoots of the common blueberry (Vaccinium myrtillus L.), herb of galega (Galega officinalis L.), grass of the mountaineer (knotweed) (Polygonum aviculare L.), roots of burdock (Arctium lappa L.), and cinnamon rose hips (Rosa cinnamomea L.) was initially investigated by reversed-phase high-performance liquid chromatography utilizing an online diode array detector and a mass spectrometer. Nine individual flavonol glycosides, derivatives of myricetin, quercetin, kaempferol, and kaempferid, were identified in HGC. The total content of flavonoids in HGC and in dry extract was determined at 6.46 and 13.96 mg/g, respectively. Robinin and kaempferol-3-glucuronide were identified as the main flavonol glycosides, the content of which in HGC was 2.09 and 2.22 mg/g, respectively.
Objectives. Herbal hypoglycemic drugs complement the conventional approach to the treatment of type-2 diabetes based on the use of synthetic prescription drugs. However, their scientifically based application and standardization are limited due to inadequate and often outdated information on their chemical composition. Accordingly, we have developed a hypoglycemic collection (HGС) consisting of common bean pods (Phaseolus vulgaris L.), bilberry shoots (Vaccinium myrtillus L.), galega herb (Gallega officinalis L.), common knotgrass herb (Polygonum aviculare L.), burdock roots (Arctium lappa L.), and cinnamon rose hips (Rosa cinnamomea L.). According to a number of researchers, the antidiabetic properties of these herbs are largely due to the presence of polyphenolic compounds, especially flavonoids. The aim of this study was to determine the profile of flavonoids in the HGС and in its total dry extract (TDE).
Methods. The study was performed by reverse-phase high-performance liquid chromatography with diode array and mass spectrometric detection.
Results. Nine individual flavonol glycosides—derivatives of myricetin, quercetin, kaempferol and kaempferide—were identified in the HGC and the TDE. The main flavonol glycosides in the studied objects were robinin and kaempferol-3-glucuronide, the contents of which in the HGC were 2.09 and 2.22 mg/g, in the TDE 4.85 and 3.84 mg/g, respectively. The other flavonol glycosides were determined in the HGC and its TDE at significantly lower concentrations.
Conclusions. The method developed in the study can be used to standardize HGCs and estimate their pharmacological activities.
SYNTHESIS AND PROCESSING OF POLYMERS AND POLYMERIC COMPOSITES
- The surface tension as well as the static and dynamic contact angles were determined for epoxy oligomers ED-20 and DER-330, laproxides 201B, DEG-1, E-181, and 703, laprolate 301, and their mixtures at different temperatures (20–60°С).
- When the content of active diluents was increased to 40 vol %, the surface tension decreased 1.5–4 times depending on the laproxide brand. The static contact angle decreased upon the introduction of active diluents into the epoxy oligomer. A 3.5-fold decrease was noted for the ED-20–laproxide 201B system at laproxide 201B content of 40 vol %. Additionally, the dynamic contact angle decreased by 15°–20° when 40 vol % of laproxides or laprolate were introduced into the epoxy oligomers.
- An increase in the temperature from 20 to 60°С led to a nearly 2-fold decrease in the surface tension for systems consisting of epoxy oligomers and active diluents. The contact angles changed by just 4°–7°.
- The impregnation rate of the fiber reinforcement containing effective binders based on mixed systems was calculated. A temperature increase in the epoxy oligomers–active diluents systems from 20 to 60°С resulted in a 10 to 20-fold impregnation rate increase.
Objectives. This study focused on the quantification of the surface tension and the static and dynamic contact angles of epoxy oligomers, active diluents, and their mixtures of various compositions at different temperatures. The active diluents were aliphatic compounds based on glycidyl ethers, namely laproxides and a laprolate of different structure, functionality, molecular weight, and viscosity. Moreover, the preparation of effective polymer binders (matrices) for composites was explored.
Methods. In this study, the epoxy oligomers ED-20 and DER-330, laproxides 201B, DEG-1, E-181, and 703, laprolate 301, and their mixtures in various compositions were investigated. Their surface tension and the static and dynamic contact angles were determined by the Wilhelmy plate and ring methods on a semiautomatic tensiometer at different temperatures (20–60 °C). The static contact angle was measured on a thin aluminum borosilicate glass plate, and the dynamic contact angles were determined using an installation for measuring surface tension developed by NPO Stekloplastik.
Results. The surface tension and static and dynamic contact angles were obtained for all epoxy oligomers and active diluents, as well as for their mixtures at 20–60 °C. For binders based on systems of epoxy oligomers and active diluents, the impregnation rate of fiber reinforcement was also calculated. The introduction of laproxides or laprolates into the epoxy oligomers led to a decrease in surface tension and contact angles, while the increase in temperature increased the impregnation rate by 10–20 times.
Conclusions. The temperature increase from 20 to 60 °C resulted in a decrease in the surface tension of mixed systems of epoxy oligomers and active diluents by almost two times. In addition, the contact angles changed by only 4°–7°, while the impregnation was significantly improved and the corresponding rate increased by 10–20 times.
CHEMISTRY AND TECHNOLOGY OF INORGANIC MATERIALS
- During the deposition of bayerite from an aluminum nitrate solution at 20°C, a precipitate with particle sizes from 0.2 to 50 μm was formed.
- Using PVA as the component of the dispersion medium for forming pastes made from bayerite powder led to a decrease in the degree of separation of colloid-bound water from the interlayer space of bayerite.
- Introduction of the η-Al2O3 powder as a heterogeneous additive in the bayerite molding paste, which was plasticized by PVA, enabled regulation of the porous structures of the resulting carriers.
Objectives. Determine the necessary conditions for obtaining a granulated η-Al2O3 carrier, investigate its structural and strength properties, and evaluate its activity for the model n-butane isomerization reaction.
Methods. Samples containing bayerite structure aluminum trihydroxide were synthesized by precipitation from aqueous solutions of aluminum nitrate with ammonia under isothermal conditions at a constant pH value. The samples of the granulated carrier were obtained using an extrusion method when the composition of molding pastes was varied by tuning the ratio of bayerite- and η-Al2O3 -containing components and introducing polyvinyl alcohol.
Results. The influence of the preparation conditions on the structural and strength properties of the active Al2O3 granules is evaluated. Samples of the aluminum oxide carrier were tested for a model reaction of low-temperature isomerization of n-butane, demonstrating a sufficiently high selectivity and reasonable prospects for use as catalysts for low-temperature isomerization of hydrocarbons.
Conclusions. Increasing the content of the polyvinyl alcohol in the molding paste from 0.4 to 1.8 wt % is accompanied by an increase in the predominant sizes of the mesopores in the range of 10–50 nm and pores in the range of 50–80 nm, explaining the high values of all recorded parameters for the process of isomerization of n-butane.
ANALYTICAL METHODS IN CHEMISTRY AND CHEMICAL TECHNOLOGY
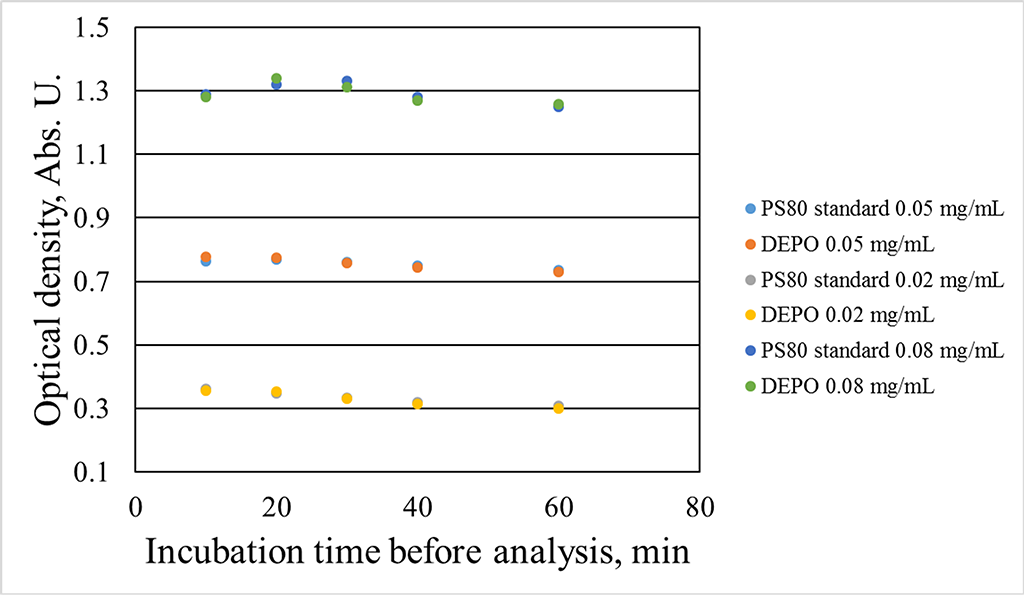
- A previously developed technique for quantification of polysorbate 80 in biopharmaceutical formulations, specifically in darbepoetin alfa and eculizumab, was enhanced.
- The conditions for each step of the analysis procedure were optimized.
- Isolation and quantification of polysorbate 80 did not depend on the matrix or, most importantly, on the protein.
Objectives. We hereby describe an improvement of a previously developed quantification technique for polysorbate 80 in biopharmaceutical formulations (darbepoetin alfa and eculizumab) and report the validation of the new approach.
Methods. Polysorbate was isolated from analyte samples by protein precipitation using an organic solvent, followed by supernatant evaporation in vacuum. Polysorbate was derivatized using a ferric thiocyanate reagent and extracted into an organic phase; the relevant optical density measurements were performed.
Results. We established the optimal conditions for each step of the analysis procedure. The accuracy was 97–102% in the tested analytical range, the relative standard deviation did not exceed 5%, and the limit of quantification was 0.01 mg/mL.
Conclusions. The reported approach is highly sensitive; polysorbate isolation and quantification do not depend on the matrix or, most importantly, the protein.
ISSN 2686-7575 (Online)